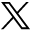Abstract
PURPOSE C-reactive-protein (CRP) is useful for diagnosis of lower respiratory tract infections (RTIs). A large international trial documented that Internet-based training in CRP point-of-care testing, in enhanced communication skills, or both reduced antibiotic prescribing at 3 months, with risk ratios (RRs) of 0.68, 0.53, 0.38, respectively. We report the longer-term impact in this trial.
METHODS A total of 246 general practices in 6 countries were cluster-randomized to usual care, Internet-based training on CRP point-of-care testing, Internet-based training on enhanced communication skills and interactive booklet, or both interventions combined. The main outcome was antibiotic prescribing for RTIs after 12 months.
RESULTS Of 228 practices providing 3-month data, 74% provided 12-month data, with no demonstrable attrition bias. Between 3 months and 12 months, prescribing for RTIs decreased with usual care (from 58% to 51%), but increased with CRP training (from 35% to 43%) and with both interventions combined (from 32% to 45%); at 12 months, the adjusted RRs compared with usual care were 0.75 (95% CI, 0.51-1.00) and 0.70 (95% CI, 0.49-0.93), respectively. Between 3 months and 12 months, the reduction in prescribing with communication training was maintained (41% and 40%, with an RR at 12 months of 0.70 [95% CI, 0.49-0.94]). Although materials were provided for free, clinicians seldom used booklets and rarely used CRP point-of-care testing. Communication training, but not CRP training, remained efficacious for reducing prescribing for lower RTIs (RR = 0.7195% CI, 0.45-0.99, and RR = 0.76; 95% CI, 0.47-1.06, respectively), whereas both remained efficacious for reducing prescribing for upper RTIs (RR = 0.60; 95% CI, 0.37-0.94, and RR = 0.58; 95% CI, 0.36-0.92, respectively).
CONCLUSIONS Internet-based training in enhanced communication skills remains effective in the longer term for reducing antibiotic prescribing. The early improvement seen with CRP training wanes, and this training becomes ineffective for lower RTIs, the only current indication for using CRP testing.
- antibiotics
- prescribing
- antimicrobial stewardship
- respiratory tract infections
- antimicrobial resistance
- primary care
- clinical practice patterns
- communication
- C-reactive protein
- practice-based research
INTRODUCTION
Acute uncomplicated lower respiratory tract infections (LRTIs) and upper respiratory tract infections (URTIs) are the leading acute presentations in primary care, and most patients still receive antibiotics1–4 despite evidence of limited benefit.4–6 Antibiotic resistance is a major public health threat, and primary care prescribing has a key role.7 Educational outreach and training in enhanced communication skills for clinicians to explore patients’ concerns can reduce antibiotic prescribing.8–13 Particular concerns for clinicians and patients are complications such as pneumonia14,15; here, C-reactive protein (CRP) point-of-care tests have utility for diagnosing LRTI.16 Physician training on use of these tests reduces antibiotic prescribing by approximately 20% in the short term,8,9 so several guidelines now advocate use of CRP in this context.17–19
Evidence for education outreach mostly documents highly expert teams helping a small number of practices, limiting generalizability of these interventions. A large pan-European trial, however, documented the impact of brief Internet-based physician training on use of a CRP point-of-care test and on enhanced communication skills using an interactive patient booklet. In the communication group, there was a clinically unimportant increase in symptom duration (a secondary outcome) of 1 day, but both interventions reduced antibiotic prescribing (the primary outcome) by 3 months, with risk ratios (RRs) of 0.53 and 0.68, respectively, and even greater benefit seen with their combination, with an RR of 0.38.20 Communication training was the most cost-effective intervention.21 It is unclear whether either of these brief interventions has longer-lasting effects on antibiotic use, which is vital to curb the emergence of antibiotic resistance. We report the impact of the interventions after 12 months.
METHODS
The methods of this trial are given in greater detail else where20; a summary is given below. Ethical approval in the United Kingdom was granted by Southampton and South West Hampshire Local Research Ethics Committee for Genomics to combat Resistance against Antibiotics for Community-acquired LRTI in Europe: INternet Training for Reducing antibiOtic use trial (short title: GRACE INTRO Trial, REC Ref 10/H0502/29). The research sites outside of the United Kingdom also obtained ethical approval from their local organizations.
Patients who met the trial’s inclusion criteria were given written and verbal information about the study and asked for informed consent. There was no individual-level consent for data collection at 12 months.
Trial Design and Audits
We chose a cluster design to minimize contamination within practices. The 4 randomization groups facilitated both 2 × 2 factorial analysis and individual group analysis.
The second audit to assess antibiotic prescribing was added after the trial began. As funding for the trial was uncertain, we asked participating clinicians for the 12-month audit only after they had completed the 3-month audit. Clinical outcomes other than prescribing were not assessed at 12 months because of resource limitations as well as the finding of minimal impact of the interventions on these outcomes at 3 months.16 For example, at 3 months, there were no significant differences between groups (described below) in hospital admissions, with 2 to 12 admissions per group, and most due to cardiorespiratory or systemic causes (eg, high fever).
Participants and Timeline
Eight primary care research networks invited local general practices to participate in the trial. The networks covered a range of countries, and their diverse health systems, languages, and cultures: England (Southampton), Wales (Cardiff), the Netherlands (Utrecht), Belgium (Antwerp), Poland (Łódź ; Szczecin), and Spain (Barcelona; semFYC). Within each practice, all clinicians who prescribed antibiotics for RTI could participate (including some UK nurse prescribers).
The trial timeline began at baseline with an audit of consecutive participants to document usual prescribing practices, conducted between October and December 2010. At the 3-month audit (February-May 2011),20 clinicians were asked to recruit 30 consecutive patients with LRTI (the main intervention target population) and 5 with URTI. At the 12-month audit (October 2011-May 2012), because the interventions had been found efficacious for both LRTI and URTI at 3 months,20 both conditions were assessed with no instructions to clinicians to preferentially recruit patients with LRTI.
Inclusion Criteria
Included practices were required to have had no prior participation in antibiotic stewardship interventions and to be able to recruit more than 10 patients at baseline.
Included patients were adults (aged 18 years or older) with LRTI or URTI. We defined LRTI as up to 28 days of cough as the most prominent symptom, or if not (eg, chills most prominent), the clinician judged the condition to be LRTI. Patients with pneumonia and chronic airway disease were included because their management could have been modified by the interventions. URTI was defined as clinician-judged other RTI (sore throat, otitis media, sinusitis, influenza, coryzal illness, or some combination thereof). Patients were excluded if they had a noninfective diagnosis (eg, pulmonary embolus), had recently received antibiotics (within the previous 28 days), were unable to provide informed consent (eg, because of dementia), were pregnant, or had immune deficiencies.
Randomization, Arms, and Interventions
Practices were remotely randomized using the minimization approach, based on practice characteristics (baseline prescribing, number of clinicians, number of patients at baseline) and with stratification by network. The 2 interventions, communication training and CRP training, were developed to be sensitive to cultural differences22 while retaining core features. Practices were randomized into 4 trial arms, as follows.
In the usual care arm, no intervention was provided. In the CRP arm, practices were given Internet-based training on use of a CRP point-of-care test. The test device was demonstrated by company representatives; the Internet training provided guidance on CRP use (Supplemental Appendix 1, available at http://www.AnnFamMed.org/content/17/2/125/suppl/DC1/). The device and testing materials were provided free. In the communication arm, practices were given Internet-based training on enhanced communication skills and use of an interactive patient booklet (Supplemental Appendix 1). The training focused on interactive use of the booklet in consultations and enhanced patient-centered communication—eliciting concerns/expectations, exchanging information, agreeing on management, summing up, and applying safety-net methods—supported by short demonstration video clips. The booklet included information about causes of symptoms, natural history, antibiotics, self-help, and when to consult the practice again. Group practices appointed a lead physician to organize a structured meeting on prescribing. In the combined intervention arm, practices received both the CRP intervention and the communication intervention.
Outcomes
Clinicians used case report forms to document symptoms and signs, and illness duration; use of CRP and booklets; and antibiotic prescribing. The trial’s primary outcome was antibiotic prescribing documented in the case report form by the recruiting clinician. Limited availability of prescription monitoring precluded use of pharmacy dispensing data.
Sample Size and Analysis
The sample size was calculated for an α of .025 and a β of .2. We assumed that 30 patients per practice would be recruited; that a 50% to 40% reduction in antibiotic prescribing would be achieved for at least 1 of the interventions8,23; and that the intracluster correlation coefficient would range from 0.168,24,25 to 0.06.26 We therefore required 2,600 patients (intracluster correlation coefficient = 0.06) to 5,400 patients (intracluster correlation coefficient = 0.16).
We used multilevel logistic regression modeling for a factorial study, controlling for baseline antibiotic prescribing rate, clustering by physician and practice, whether the patient had a URTI or LRTI, and a range of potential confounders. There was no additional effect of international network; hence, it was not included in models. We undertook a secondary analysis for the individual randomization groups because the study was not powered for interactions. The odds ratios were converted to RRs.27 Analysis was conducted on an intention-to-treat basis. To assess attrition bias (1) we used data aggregated at the practice level, and multiple imputation to impute data from practices that had not agreed to be followed up, and (2) we compared estimates at 3 months just for those practices followed up at 12 months.
RESULTS
Participants and Follow-up
At the baseline audit, 5,355 patients (79.1%) had LRTI and 1,416 (20.9%) had URTI. Overall, 3,742 (55%) were prescribed antibiotics.
A total of 372 participating clinicians in 228 of 246 practices contributed data for 4,264 patients at the 3-month follow-up (Figure 1), of whom 20% had URTI. Of the 228 practices providing 3-month data, 168 (74%) provided 12-month data. A total of 247 clinicians in these 168 practices contributed data for 4,830 patients at the 12-month follow-up, of whom 41% had URTI; hence, we controlled for URTI in the estimates.
CONSORT flow diagram.
CONSORT =Consolidated Standards of Reporting Trials; CRP = C-reactive protein.
The groups were well balanced at 3 months and remained so at 12 months (Table 1). Initial compliance with training was good, with most clinicians completing all modules: CRP, 99 of 113 (88%); communication, 94 of 108 (87%); and combination, 116 of 127 (91%).
Patient Characteristics
Use of Study Materials
By 12 months, clinicians in all groups had seldom used CRP testing in patient care, even though they were given free access to CRP diagnostic kits: usual care, 16 of 1,195 (1.34%); CRP, 62 of 1,075 (5.77%); communication, 56 of 1,168 (4.79%); and combination, 85 of 1,419 (5.99%). Booklets were used fairly sparingly too: communication, 189 of 1,186 (16%) and combination, 340 of 1,428 (24%).
Efficacy of the Interventions
Factorial Analysis
Results of factorial analysis showed that at 3 months, 48% (984 of 2,040) of the patients consulting clinicians who were not trained in using CRP were prescribed antibiotics, and by 12 months, the value was little changed, at 46% (1,078 of 2,360) (Table 2). The antibiotic prescribing rate in the CRP groups was 33% (734 of 2,224) at 3 months but 45% (1,097 of 2,462) at 12 months; however, the adjusted RR was a nonsignificant 0.87 compared with the no-training control at 12 months (95% CI, 0.68-1.06; P = .18).
Factorial Analysis: Efficacy of Interventions in Reducing Antibiotic Prescribing at 12 Months
At 3 months, 45% (876 of 1,932) of the patients consulting clinicians who were not trained in enhanced communication were prescribed antibiotics, and similarly, at 12 months, 48% (1,069 of 2,246) were. The antibiotic prescribing rate in the communications groups was 36% (842 of 2,332) at 3 months but 43% (1,106 of 2,576) at 12 months; the adjusted RR compared with no training at 12 months was a significant 0.81 (95% CI, 0.64-1.00; P = .049).
Analysis of Individual Groups
The factorial analysis probably masks effectiveness of individual interventions because there was a sizable interaction term between the CRP and communication interventions (1.67; P = .16). The individual group analysis yielded somewhat different results (Table 3).
Individual Group Analysis: Efficacy of Interventions in Reducing Antibiotic Prescribing at 12 Months
Among patients receiving usual care, whose clinicians received no training, 58% (508 of 870) were prescribed antibiotics at 3 months, and the value had decreased by an absolute 7%, to 51% (613 of 1,194), at 12 months in part due to more cases of URTI. Prescribing in the CRP group, however, rose by almost 9% from 35% (368 of 1,062) at 3 months to 43% (456 of 1,052) at 12 months (adjusted RR compared with usual care at 12 months = 0.75; 95% CI, 0.51-1.00; P = .052). Similarly, among patients in the combined intervention group, 32% (476 of 1,170)were prescribed antibiotics at 3 months, with an absolute 13% increase to 45% (641 of 1,410) at 12 months (adjusted RR compared with usual care at 12 months = 0.70; 95% CI, 0.49-0.93; P = .01). Among those in the communication group, 41% (476 of 1,170) were prescribed antibiotics at 3 months, with virtually no change by 12 months, 40% (465 of 1,166) (adjusted RR compared with usual care at 12 months = 0.70; 95% CI, 0.49-0.94; P = .02).
Results differed somewhat by type of RTI. For patients with LRTI, communication was still effective compared with usual care at 12 months (adjusted RR = 0.71; 95% CI, 0.45-0.99), but CRP was not (adjusted RR = 0.76; 95% CI, 0.47-1.06). For patients with URTI, benefit of both interventions relative to usual care was maintained at 12 months: communications (adjusted RR = 0.60; 95% CI, 0.37-0.94; P = .02) and CRP (adjusted RR = 0.58; 95% CI, 0.36-0.92; P = .02).
Assessment of Attrition Bias
Data from practices where follow-up was possible were comparable to those for the entire trial population (Supplemental Appendix 2, Supplemental Table 5, and Supplemental Table 6, available at http://www.AnnFamMed.org/content/17/2/125/suppl/DC1/). In our practice-level analysis, we compared results for the practices completing follow-up at 12 months with estimates for all practices that were derived with imputation for missing data (Table 4). Although the absolute estimates of efficacy differed slightly, with lower power and less robustness (because of inability to control for individual patient characteristics), there was no meaningful change in the practice-based estimates. This finding suggests that there was minimal attrition bias related to practices not completing the study.
Assessment of Attrition Bias: Completing Practices and All Practices With Imputation
As a further check, we determined that estimates at 3 months from completers were also very similar to results for the whole trial cohort. The RRs were 0.54 for CRP and 0.68 for communication among completers, and 0.54 and 0.69, respectively, among the whole trial cohort.
DISCUSSION
As far as we are aware, this is the first major multi-center international trial to assess longer-term effectiveness of Internet-based training to modify antibiotic prescribing for RTIs. Although antibiotic prescribing decreased with usual care during follow-up out to 12 months, communication training remained more efficacious at that time, whereas CRP training did not.
Potential Limitations
Our trial had a number of potential limitations. Practices were approached only after 3 months to request further follow-up, and 26% of those providing 3-month data declined to continue. Baseline characteristics, however, were similar for practices that were not followed up, and the practice-level analysis using multiple imputation demonstrated no attrition bias. The majority of practices approached agreed, and many of them had not previously taken part in research.
As expected, clinicians reported struggling to document consecutive patients at busy times of year. But previous research has shown this challenge results in little selection bias,28,29 and there were minimal barriers to participation for the 12-month audit (the trial used quick audit proformas, and there was no delay for patient consent). Antibiotic prescribing was similar to that seen in previous studies,2,4 and at baseline, the majority of patients in usual care received antibiotics, which suggests the trial’s results are generalizable.
There was no evidence of differential selection bias comparing groups, and analysis controlled for differences in case mix. At 12 months, usual care prescribing had fallen slightly, by 7%, perhaps because of pressure on clinicians (eg, the European Antibiotic Awareness Day), but also, there were more patients with URTI at that time point, which explained one-half the reduction. The findings cannot be explained by case mix, however, because we controlled for a range of variables in the analysis. We checked fidelity with initial training but did not observe actual consultations (to avoid influencing behavior). We did not record other clinical outcomes at 12 months given the modest impact seen at 3 months.20
Comparison With Other Studies
In this trial, the benefit of communication training was durable, supporting prior evidence for interactive methods.10,11 Our process evaluation also indicated that communication training promoted changes in physician attitudes that should be helpful in the longer term.22,30,31 The Stemming the Tide of Antibiotic Resistance (STAR) trial achieved a 4% reduction in overall antibiotic prescribing but was more intensive (5 online phases vs 1 in this study, plus expert-led outreach seminars).13 Our intervention yielded less initial reduction than the Dutch IMPAC3T trial,8 but that trial was also more intensive, with face-to-face communication training. The long-term effect on behavior in our study was similar to that seen with other, more intensive interventions.8,13,32 Booklets were initially important, but their limited use in follow-up—despite the intervention’s persistent efficacy—suggests that clinicians had consolidated skills and were being more selective in using this resource.
Communication was slightly less effective than CRP training or the combined intervention at 3 months (RRs = 0.68 vs 0.53 and 0.38, respectively), a finding comparable to the trend seen in the Dutch trials.8,9 Even though all materials were provided for free, CRP tests were little used by 12 months, and the effectiveness of CRP training had waned substantially (RRs = 0.70, 0.75, and 0.70, respectively)—but the diminished impact was particularly pronounced among patients with LRTI, where use of CRP to guide prescribing is supported.17–19 Our findings support long-term findings for LRTI from the smaller, intensive IMPAC3T trial, where there was no longer a significant effect of CRP training at follow-up, but the effect of communication training persisted.32 The waning in efficacy of CRP training may reflect the general waning of quality improvement interventions over time, although no similar effect occurred with communication training. It is unclear whether further reduction in the efficacy of CRP training would occur beyond 12 months. The logistics of providing this training and the time required to do the test at the busiest times of year may also be key disincentives to longer-term engagement.
The persistent benefit of CRP training in reducing antibiotic prescribing for URTI despite low use of CRP may be due to clinicians having learned to prescribe fewer antibiotics when using CRP, or to the training content for both CRP and communication, which shared introductory modules about the limited benefit of antibiotics, that is, that they have a nonspecific effect that is possibly unrelated to CRP per se. Ongoing incentives would probably be needed for clinicians to continue using CRP testing, but evidence of cost-effectiveness of this approach would be needed. As communication training was the most cost-effective option at 3 months when allowing for the costs of antibiotic resistance,21 its lasting impact makes it likely to be even more cost-effective.
Conclusion
Internet-based training in enhanced communication skills to reduce antibiotic prescribing for RTI remains efficacious in the longer term. In contrast, an early improvement with CRP training wanes over time, and this strategy becomes ineffective long term both overall and for LRTI, the only current indication for using CRP to guide decisions about antibiotic therapy. Our trial’s findings suggest there is probably only short-term benefit from training clinicians in routine primary care to use CRP. Instead, the most useful training for long-lasting effects is likely in enhanced communication skills.
Acknowledgments
We are grateful to members of the GRACE consortium trial team whose hard work has made this possible. We are very grateful to the clinicians and patients who consented to be part of GRACE, without whom this study would not have been possible. We also thank the independent GRACE trial steering committee (Patrick Bindels, Gordon Taylor, and Mark Woodhead) for their help and suggestions. The trial steering committee also supervised safety aspects of the trial as there was no independent data safety monitoring board (following similar precedents with low-risk MRC-funded trials, as the study was considered to be low risk). We are grateful to Orion Diagnostica for supplying all CRP materials.
Footnotes
Conflicts of interest: authors report none.
Author contributions: The initial idea for this study was proposed by P.L., T.V., C.C.B, S.C., and H.G.; all authors contributed to the development of the protocol, and to the management of the study; G.O. and C.B. led the day-to-day management of the study supervised by P.L. and T.V., respectively; L.Y. led and supervised the design and development of the web-based intervention; L.Y., N.F., P.L., and C.C.B. led the development of the communication intervention. J.C., H.M., and P.L. led the development of the CRP intervention. Both interventions benefited from input of the network leads. Frank Leus supervised the web-based data management. H.G. led the funding application and provided overall coordination of the GRACE consortium; K.H. coordinated randomization, and M.K., P.L., B.S., and M. Mullee analyzed the data; all authors contributed to the interpretation of the data and the write-up. P.L., M. Mullee, and B.S. had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Funding support: The research performed by the GRACE (Genomicsto combat Resistance against Antibiotics in Community-acquired LRTIin Europe) consortium leading to these results was funded by the European Community’s Sixth Framework Programme under grant agreement no. 518226. The work in the United Kingdom was also supported by the National Institute for Health Research (NIHR) (service support costs) and by the Research Foundation–Flanders (G.0274.08N). Orion Diagnostica supplied all the equipment and consumables for CRP testing.
The work reported as part of the final follow-up was also supported through the European Science Foundation (ESF), in the framework of the Research Networking Programme TRACE (www.esf.org/trace). Contributors to this programme: Bond University (Australia); Research Foundation–Flanders, University of Antwerp, University of Ghent (Belgium); Chinese University of Hong Kong (China); University of Copenhagen (Denmark); Research Council of Health, Academy of Finland (Finland); College Azuréen des Généralistes Enseignants, Comité Départemental d’Education pour la Santé (France); Rostock University (Germany); The Netherlands Organisation for Scientific Research, AMC Amsterdam, Leiden UMC, UMC Utrecht (The Netherlands); Research Council of Norway, University of Oslo, University of Tromso (Norway); Medical University of Bialystok, Medical University of Lodz (Poland); National University Research Council (Romania); Osnovno zdravstvo Gorenjske (Slovenia); l’Institut d’Investigacions Biomèdiques August Pi i Sunyer (Spain); Swedish Research Council, Karolinska Institute (Sweden); Medical Research Council, Cardiff University, University of Oxford, University of Southampton (United Kingdom); Swiss National Science Foundation (Switzerland).
Disclaimer: The funders and sponsor (University of Southampton) had no role in the management, analysis, interpretation or write-up of this project. The principal investigator (P.L.) made the decision to submit for publication following agreement from all authors.
Trial Registration: ISRCTN99871214
Supplementary materials: Available at http://www.AnnFamMed.org/content/17/2/125/suppl/DC1/.
- Received for publication July 30, 2018.
- Revision received December 10, 2018.
- Accepted for publication December 31, 2018.
- © 2019 Annals of Family Medicine, Inc.